Motor Health — Medical Devices
Powered by Data Management for Edge AI
In modern medical devices, motors are no longer simple electromechanical components. They are precision actuators responsible for life-critical motion, fluid control, positioning, and thermal management. From infusion pumps to surgical robots, ventilators to imaging systems, motor reliability directly impacts patient safety, regulatory compliance, and device uptime.
Medical devices and medical robotic arm systems must process and manage critical data directly within the device to ensure safety, precision, and regulatory compliance. These systems continuously generate operational data from sensors measuring position, force, torque, vibration, temperature, and system telemetry, which must be captured, processed, and stored in real time. Because surgical and clinical environments require deterministic behavior, low latency, and complete traceability, relying solely on external systems or cloud connectivity is not acceptable.
By managing data locally, medical robotics platforms can support real-time control decisions, continuous monitoring, fault detection, and full audit trails of device behavior. This on-device data management is essential for patient safety, explainable system actions, and compliance with medical regulations, while enabling advanced capabilities such as AI-assisted procedures and predictive maintenance of critical components.
In addition, to ensure safe, explainable, and deterministic motor intelligence at the device level, manufacturers need more than AI models. They need structured, power-fail-safe, real-time data management at the edge.
That is where the ITTIA DB Platform transforms motor health in medical systems.
Why Motor Health Matters in Medical Devices
Effective data management is essential for monitoring motor health in medical devices because many critical systems rely on motors for precise and reliable operation. Devices such as robotic surgical arms, infusion pumps, imaging systems, ventilators, and laboratory automation platforms depend on motors to deliver controlled movement, fluid delivery, and positioning with high accuracy. These motors continuously generate operational signals, such as current, vibration, temperature, torque, and speed, that provide valuable insight into their condition.
Proper data management ensures these signals are captured, structured, stored, and analyzed reliably within the device, enabling early detection of issues like bearing wear, overheating, misalignment, or abnormal load conditions. By preserving this data and making it traceable, medical systems can support real-time monitoring, predictive maintenance, and post-event analysis, while also meeting strict safety, reliability, and regulatory requirements.
Medical motors operate under strict constraints:
- Continuous or high-duty cycles
- Tight torque and speed tolerances
- Silent, vibration-controlled environments
- Long service lifetimes
- Zero tolerance for unexpected failure
Examples include:
- Infusion pumps (flow precision)
- Ventilators (airflow control)
- Surgical robotics (micro-positioning)
- Lab automation systems
- Imaging equipment cooling and positioning systems
A minor anomaly in vibration, current draw, or thermal behavior can indicate early wear. If captured correctly, these signals enable:
- Predictive maintenance
- Drift detection
- Regulatory traceability
- Post-incident root-cause analysis
But only if the data is captured deterministically and preserved with integrity.
Ultimately, strong on-device data management helps ensure that motor-driven medical devices operate safely, consistently, and transparently in patient-critical environments.
The Missing Layer: Deterministic Edge Data Management
AI models alone cannot ensure safe motor monitoring. The real foundation is structured, time-series data management at the device level which refers to the practice of organizing and storing continuously generated device data, such as sensor readings, system states, and operational metrics, in a well-defined, queryable format directly on the embedded device. Instead of treating incoming signals as temporary streams or simple log files, the data is captured as timestamped records arranged in time order, forming a time-series that represents how the system behaves over time. This structured approach allows embedded systems to efficiently store, retrieve, analyze, and correlate historical data, making it possible to detect trends, generate feature windows for AI models, and trace system behavior back to specific events. By managing time-series data locally on the device, systems gain reliable observability, faster access to operational history, and the ability to perform analytics or AI inference without relying on constant cloud connectivity. This is particularly important for applications such as robotics, industrial automation, medical devices, and predictive maintenance, where understanding how signals evolve over time is critical for reliability and decision-making.
Without this:
- Data becomes fragmented
- Flash wear becomes unpredictable
- Latency spikes occur
- Explainability disappears
- Regulatory traceability is lost
The ITTIA DB Platform delivers this foundation.
The ITTIA DB Platform for Medical Motor Intelligence
ITTIA DB Lite — Deterministic Data on MCUs
Designed for microcontrollers inside medical devices, ITTIA DB Lite provides:
- Deterministic time-series ingestion
- Power-fail-safe storage
- Controlled flash erase behavior
- Background clean-block management
- Wear-leveling awareness
- Structured storage for signals, features, and health metrics
What this enables:
- Reliable capture of motor current, vibration, temperature, and speed
- Fixed worst-case latency (WCET predictable)
- No blocking writes during erase
- Compliance-ready traceability logs
- AI-ready feature windows stored locally
This is critical for real-time devices where latency and predictability directly affect safety.
ITTIA DB — Structured Intelligence on MPUs
For Linux/QNX-based medical systems, ITTIA DB provides:
- Embedded relational + time-series database
- Structured logging for motor diagnostics
- Local model performance tracking
- Data versioning for compliance
- Secure persistence for long operational lifetimes
It allows:
- Storing sensor → signal → feature → inference → action chain
- Recording model versions tied to motor behavior
- Tracking anomaly scores over time
- Enabling post-event forensic analysis
In regulated environments, explainability is not optional, it is mandatory.
ITTIA Analitica — Visualization & Insight
Motor intelligence must be understandable by engineers, QA teams, and field technicians.
ITTIA Analitica provides:
- Local or fleet-level visualization
- Time-series dashboards
- Drift analysis
- Health scoring trends
- Model accuracy tracking
- Comparative fleet behavior insights
This helps manufacturers:
- Validate predictive maintenance models
- Identify systemic wear patterns
- Detect calibration drift
- Optimize firmware + AI updates
ITTIA Data Connect — Secure Fleet Synchronization
Medical devices operate in distributed environments, hospitals, labs, clinics.
ITTIA Data Connect enables:
- Secure data synchronization
- Selective export of structured motor data
- Bandwidth-efficient replication
- Model improvement feedback loops
Instead of sending raw sensor streams, devices export:
- Cleaned, structured feature windows
- Health metrics
- Anomaly summaries
- Model performance telemetry
This reduces cloud costs while preserving compliance integrity.
The Edge AI Motor Pipeline in Medical Devices
Here’s how it works end-to-end:
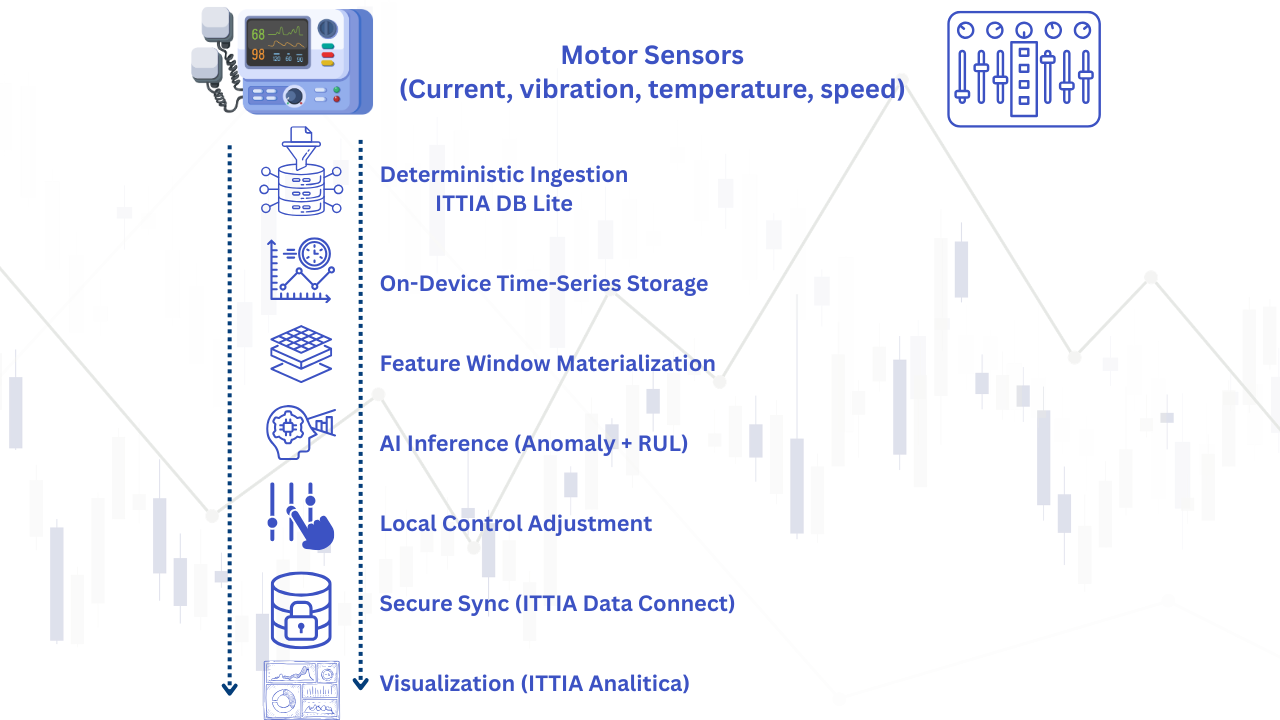
This pipeline ensures:
- No data loss
- No unpredictable latency
- No flash-related stalls
- Full traceability
- Production-grade Edge AI
Regulatory & Safety Alignment
Medical devices face standards such as:
- IEC 62304 (software lifecycle)
- ISO 14971 (risk management)
- IEC 60601 (medical electrical safety)
- Cybersecurity requirements
- AI explainability expectations
The ITTIA DB Platform supports conformance by:
- Enabling deterministic behavior
- Maintaining structured logs
- Preserving historical context
- Recording model + firmware versions
- Supporting audit trails
AI without structured data is fragile. AI with deterministic data becomes explainable and compliant.
Why This Matters Now
Medical systems are increasingly evolving toward higher device autonomy, AI-assisted control loops, reduced downtime, remote service models, and predictive maintenance capabilities. In many of these systems, motors are the first mechanical components to show signs of degradation, as they continuously perform precision motion and actuation. By capturing early indicators of wear—such as changes in current, vibration, temperature, or torque, in a safe and deterministic manner, medical devices can detect issues long before they impact performance or patient safety. This shift in data awareness transforms traditional reactive repair into predictive intelligence, enabling systems to anticipate failures and schedule maintenance proactively. Ultimately, it leads to more autonomous, reliable medical devices that maintain precision and availability in critical healthcare environments.
Motor Intelligence at the Medical Edge
The future of medical devices is not defined only by smarter algorithms, but by data-centric Edge AI that manages and understands device behavior directly within the system. In this model, every motor signal is captured deterministically, every anomaly can be explained, every decision is traceable, and every device contributes meaningful insights to fleet-wide intelligence. The ITTIA DB Platform, comprising ITTIA DB Lite, ITTIA DB, ITTIA Analitica, and ITTIA Data Connect, provides a complete, production-grade data foundation that enables reliable motor health intelligence in medical devices. By ensuring structured, persistent, and observable data from signal capture through analytics and telemetry, the platform helps medical systems operate with transparency and reliability. Because in healthcare, unpredictability is unacceptable, deterministic data is safety.